The American Association for the Study of Liver Disease (AASLD), the organization that defines how doctors should treat hepatitis B and other liver ailments, unveiled new hepatitis B treatment guidelines this week at its annual conference in San Francisco The new guidelines are published here Patients should review them and discuss any updates that address theirCROI Hepatitis, HCV, HBV & Fatty Liver at CROI (04/29/) AASLD Hepatitis B reactivation after interferonbased therapy versus panoral direct acting antiviral agents in chronic hepatitis C patients coinfected with hepatitis B virus a systematic review and metaanalysis () AASLD Development of Second Generation RNA Interference Therapy for Hepatitis BProgram at a Glance 6 Saturday, November 14 Time (ET) Sessions Flipped Learning Course Postgraduate Course Paradigm Shifts in Hepatology Rethinking Conventional Wisdom 900 – 1030 am AASLD/Japan Society of Hepatology Joint Symposium Lean NAFLD Emerging Trends Symposium Artificial Intelligence (AI) in Hepatology
Www Aligos Com Wp Content Uploads 11 Development Of A Best In Class Hbv Aso Alg 0572 For The Treatment Of Chronic Hepatitis B Pdf
Aasld hepatitis b 2020
Aasld hepatitis b 2020-1University of California San Francisco, San Francisco, CA 2Ochsner Medical Center, New Orleans, LA 3Corporal Michael J Crescenz VA Medical Center & University of Pennsylvania Perelman School of Medicine, Philadelphia, PA 4The University of Texas MD Anderson Cancer Center, Houston, TX 5Boston Children's Hospital, Harvard Medical School,(4) monitoring of untreated patients;




Hbv Drugs In Development Hepatitis Clinical Care Options
Dicerna Announces Updated Phase 1 Data on RG6346 Investigational Treatment for Chronic Hepatitis B Virus to be Presented in LateBreaking Session at AASLD's The Liver Meeting ®Guidance for design and endpoints of clinical trials in chronic hepatitis B Report from the 19 EASLAASLD HBV Treatment Endpoints Conference ‡ J Hepatol Mar;72(3) doi /jjhep3 Epub 19 Nov 12 Authors Markus Cornberg 1 , Anna SukFong Lok 2 , Norah A Terrault 3 , Fabien Zoulim 4 , 19 EASLAASLD HBV Treatment Endpoints Conference(AASLD) on the treatment of chronic hepatitis B (CHB) virus (HBV) infection in adults and children Unlike previous AASLD practice guidelines, this guideline was developed in compliance with the Institute of Medicine standards for trustworthy practice guidelines and uses the Grading of Recommendation Assessment, Development
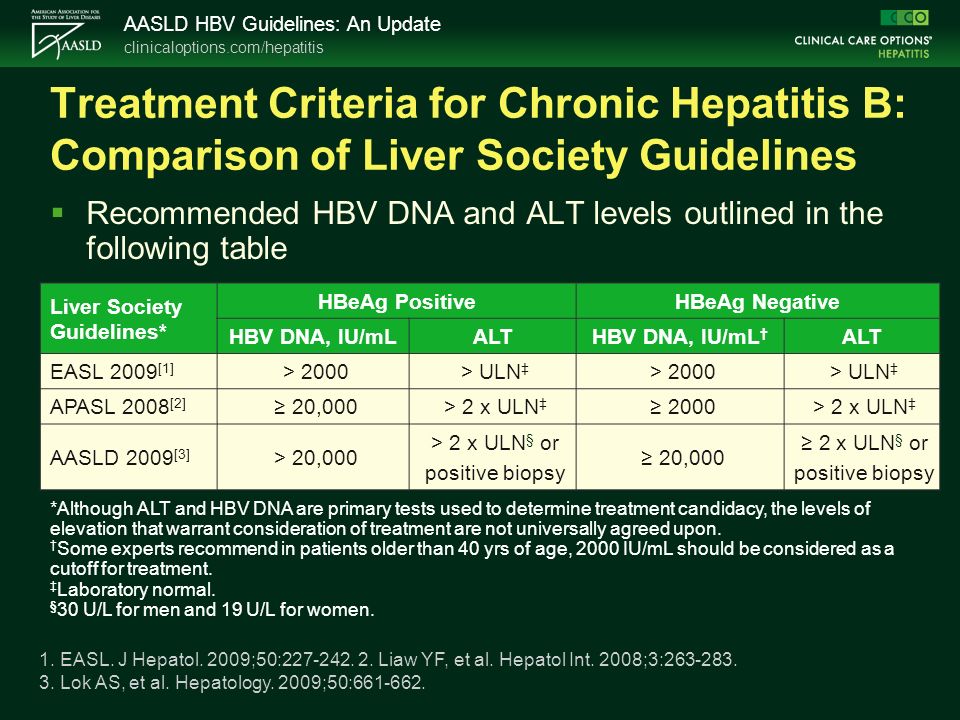
The HBV Primary Care Workgroup Guidance was first released in early and is accessible on this web site (Hepatitis B Online), with the aim to have regular updated versions posted online The goal of this document is to provide simplified, uptodate, and readily accessible HBV management guidance for primary care medical providers Chronic hepatitis B HBsAg positive for more than six months, serum HBV DNA greater than ,000 IU per mL (lower values of 2,000 to ,000 IU per mL often occur with HBeAgnegative chronicAASLD 19 Triple Therapy With Lonafarnib, Ritonavir, and PegInterferon Lambda 1a Appears Promising for Hepatitis Delta Virus Infection PracticeUpdate Editorial Team AASLD AASLD 19 Transient Elastography May Help in the Assessment of Allograft Quality in Deceased Liver Donors PracticeUpdate Editorial Team AASLD AASLD
Lisa Catalli is a Nurse Practitioner at the University of California, San Francisco's Viral Hepatitis Center and Liver Transplant Clinic She earned her Master's Degree from UCSF School of Nursing in 1995 and has been working in the field of Hepatology since 10 She has been treating HCV since the dawn of directacting antiviral therapy, and served as a coinvestigator in several Guidance for design and endpoints of clinical trials in chronic hepatitis B—report from the 19 EASLAASLD HBV Treatment Endpoints Conference J Hepatol ;72(3) doi /jjhep3 PubMed Google Scholar CrossrefDoylestown, Pa, Nov 12, – Hepatitis B Foundation cofounders Joan Block, RN, and Timothy M Block, PhD, will receive a very significant honor on Nov 14 during The Liver Meeting, which is the annual meeting of the American Association for the Study of Liv er Diseases (AASLD)



Hepatitis B Chapter 4 Yellow Book Travelers Health Cdc




Core Concepts Hepatitis B Coinfection Co Occurring Conditions National Hiv Curriculum
Multiple Abstracts Highlighting Assembly Biosciences' Hepatitis B Core Inhibitor Development Programs Accepted for Presentation at the AASLD The Liver Meeting Digital Experience™ read this article along with other careers information, tips and advice on BioSpace McMahon has served as coauthor of the AASLD Practice Guideline for Hepatitis B between 09 and 18 and is coAuthor of the AASLD Hepatitis B Guidance 19 He was also the cochair of the WHO Hepatitis Guideline published in 15 He directed a program that halted transmission of hepatitis B in the Alaska Native population in the 1980's, the only US born population endemic for persons with chronic hepatitis BAASLD encourages you to make the most out of your member benefits Discounted Member rates are benefits offered to AASLD members when you register for The Liver Meeting To qualify, membership must be current at the time of registration Please take a moment to renew your membership Thank you and we look forward to seeing you at The Liver Meeting Digital




Hepatitis B Foundation Founders Joan And Tim Block Receive Advocacy Award
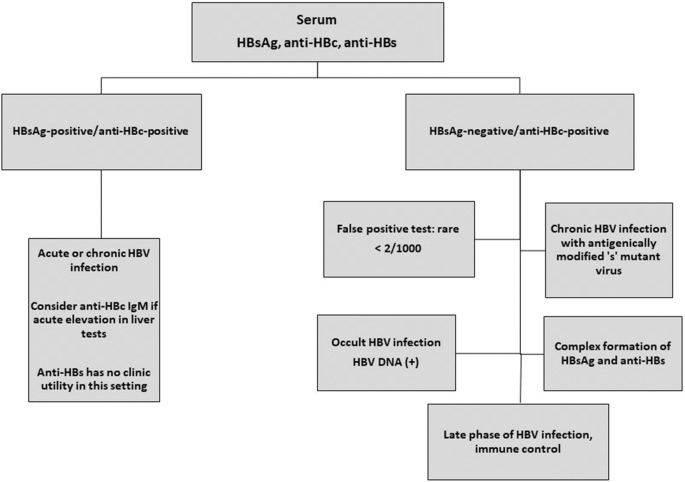



Hepatitis B Core Antibody Role In Clinical Practice In Springerlink
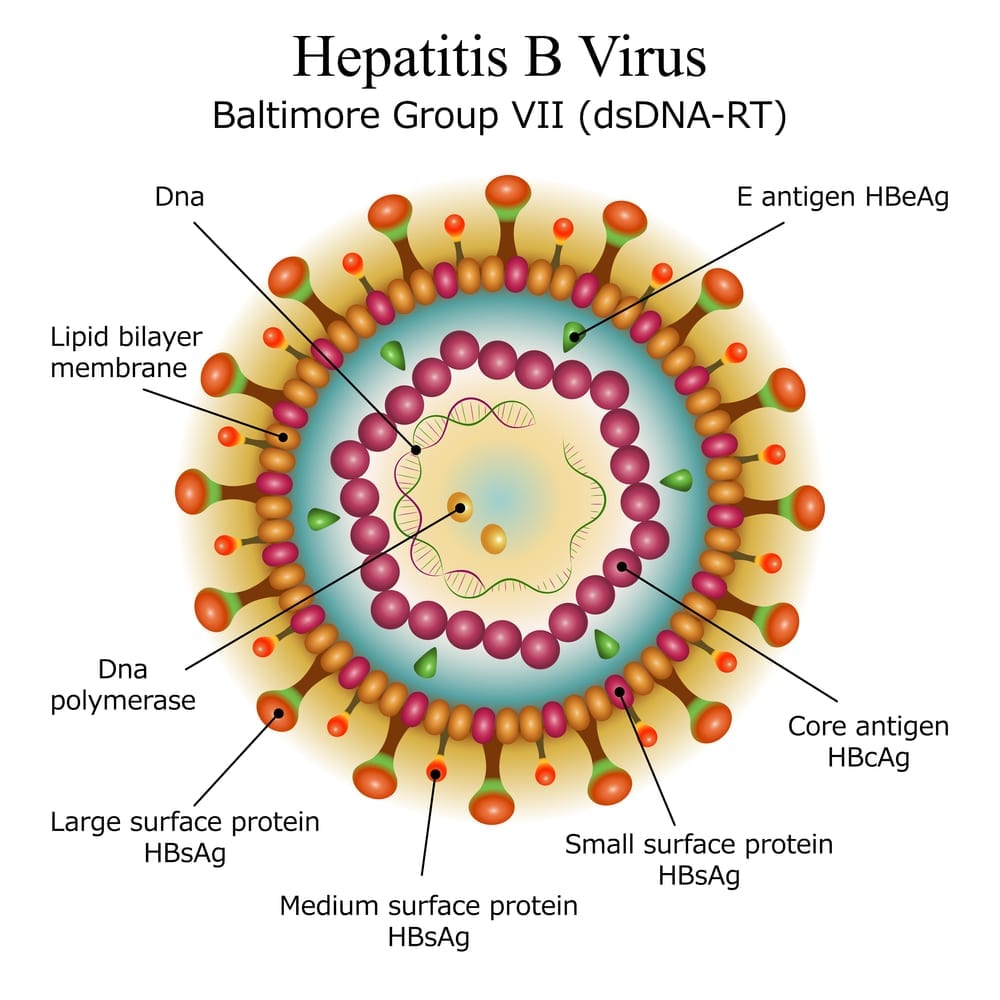
Hepatitis B virus (HBV), a small, circular, partially doublestranded DNA virus in the family Hepadnaviridae TRANSMISSION HBV is transmitted by contact with contaminated blood, blood products, and other body fluids (such as semen) Examples of exposures associated with transmission that travelers may encounter include poor infection control during medical or dental Webinar AASLDALEH COVID19 and Liver Disease in the Americas – 21 and Beyond Webinar Mental Health & Burnout in the Pandemic Name It, Frame It and Tame It Emerging Topic Conference Chronic Hepatitis B From the Population to New Molecules and Back Webinar Simultaneous Liver Kidney Transplant How Did We Do With the New Criteria?(3) specialized virological and serological tests;




News Hepatitis Australia




Comparison Of Clinical Practice Guidelines For The Management Of Chronic Hepatitis B When To Start When To Change And When To Stop
Emerging Topic Conference Chronic Hepatitis B From the Population to New Molecules and Back AASLD / FDA DILI Conference Druginduced Liver Injury New Developments and Innovations in Patients with Underlying Liver Disease, Cancer or COVID19 Digestive Disease Week Transplant Hepatology Board Review Course The HCC screening method recommended in the 18 AASLD Hepatitis B Guidance is ultrasonography every 6 months, Hepatocellular carcinoma prediction beyond year 5 of oral therapy in a large cohort of Caucasian patients with chronic hepatitis B J Hepatol Jan 22Epub ahead of print PubMed Abstract 36 Momin BR, Pinheiro PS, Carreira H, Li C, WeirAnd (5) treatment of hepatitis B in special populations, including persons with viral coinfections, acute hepatitis B




Cldf




Guidance For Design And Endpoints Of Clinical Trials In Chronic Hepatitis B Report From The 19 Easl sld Hbv Treatment Endpoints Conference Journal Of Hepatology
Emerging Topic Conference Chronic Hepatitis B From the Population to New Molecules and Back AASLD / FDA DILI Conference Druginduced Liver Injury New Developments and Innovations in Patients with Underlying Liver Disease, Cancer or COVID19 Digestive Disease Week Transplant Hepatology Board Review Course Currently, there is groundbreaking research on HBV cure efforts, therapeutic developments for HDV, virologic biomarkers to help individualize antiviral "stopping" strategies in selected populations, and our understanding of concurrent NAFLD and chronic HBV infection This twohour symposium reviews emerging data on a potential HBV cure and HDV Dicerna Announces Updated Phase 1 Data on RG6346 Investigational Treatment for Chronic Hepatitis B Virus to be Presented in LateBreaking Session at AASLD's The Liver Meeting® Digital Experience™ read this article along with other careers information, tips




Hepatitis B Amboss




Cldf
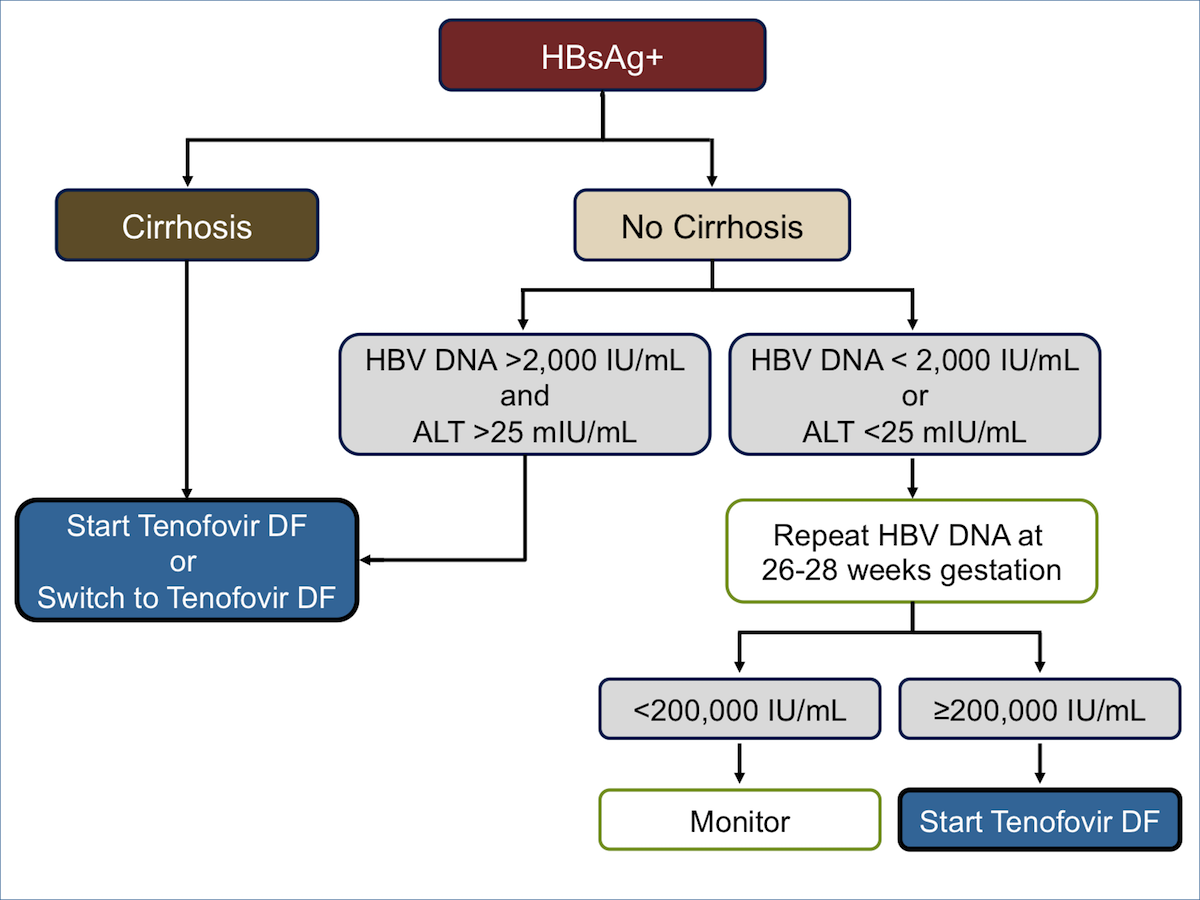
Her translational research focuses on immune pathogenesis in human hepatitis C and B virus infection with and without HIV coinfection—including various immune regulatory mechanisms such as FoxP3 Tregs, IL10 Tr1 cells, gdTcells, PD1 and CTL Dr Chang is also participating in the Million Veteran Program—a multicenter VA genomic study She is a Fellow ofAntiviral therapy in chronic hepatitis B viral infection during pregnancy A systematic review and metaanalysis(link is external) Website December 19 Hepatitis C Guidance 19 Update AASLD‐IDSA Recommendations for Testing, Managing, and Treating Hepatitis C Virus InfectionThe 18 updated guidance on chronic hepatitis B (CHB) includes (1) updates on treatment since the 16 HBV guidelines (notably the use of tenofovir alafenamide) and guidance on (2) screening, counseling, and prevention;




Viral Hepatitis B In Pregnancy Hdv The Nepali Doctor



Www Aligos Com Wp Content Uploads 11 Development Of A Best In Class Hbv Aso Alg 0572 For The Treatment Of Chronic Hepatitis B Pdf
Webinar AASLDALEH COVID19 and Liver Disease in the Americas – 21 and Beyond Webinar Mental Health & Burnout in the Pandemic Name It, Frame It and Tame It Emerging Topic Conference Chronic Hepatitis B From the Population to New Molecules and Back Webinar Simultaneous Liver Kidney Transplant How Did We Do With the New Criteria? The use of immunosuppressive medications in people with hepatitis B virus (HBV) infection is associated with an increased risk of HBV reactivation, which can lead to liver failure and death Tumour necrosis factor inhibitors, rituximab and other biologic treatments have been associated with HBV reactivation in up to 24% of people with resolved HBV (positive core• Consider etiologies unrelated to COVID19, including other viruses such as hepatitis A, B and C, and druginduced liver injury when assessing patients with COVID19 and elevated liver biochemistries • To limit unnecessary transport of patients with COVID19, ultrasound or other advanced imaging



Guidance For Design And Endpoints Of Clinical Trials




Clinicalthought Hbv Treatment Advances Clinicalthought sld Hepatitis Clinical Care Options
Omar Massoud, MD, PhD, FAASLD is an associate professor of medicine at the University of Alabama at Birmingham He is the director of clinical liver research at the University of Alabama Liver Center He is a member of the AASLD Publication Committee and Hepatitis C Special Interest Group Steering Committee He earned his Doctor of Medicine degree from Al Aligos Therapeutics Presents Data on Chronic Hepatitis B, NASH and Hepatocellular Carcinoma Programs at AASLD's The Liver Meeting Digital Experience™ Dicerna Announces Positive Updated Data From Phase 1 Trial of RG6346 for Treatment of Chronic Hepatitis B Virus (HBV) Infection at AASLD's The Liver Meeting ® Digital Experience™




sld 19 Assembly Asks Investors To Keep The Faith Evaluate




sld Guidelines Hepatitis B 18 Ppt
A National Strategy for the Elimination of Hepatitis B and C In the face of recent developments in hepatitis C treatment, enhanced global solidarity for hepatitis B and hepatitis C elimination goals, and progress implementing The National Viral Hepatitis Action Plan (17) pdf icon PDF – 84 pages external icon, an ambitious strategy to set and achieve HBV and HCV elimination goals Serologies for hepatitis B should include HBsAg, hepatitis B surface antibody (antiHBs), total hepatitis B core antibody (antiH), and IgM antibody to hepatitis B core antigen (IgM antiH) Those who do not have HBV infection should be offered hepatitis B vaccination series according to the ACIPrecommended scheduleHIVandHepatitiscom coverage of the 17 American Association for the Study of Liver Diseases (AASLD) Liver Meeting in Washington, DC, October 24, 17 Conference highlights include directacting antiviral therapy for difficulttotreat people with hepatitis C, novel hepatitis B agents, complications of viral hepatitis, and NAFLD/NASH



3
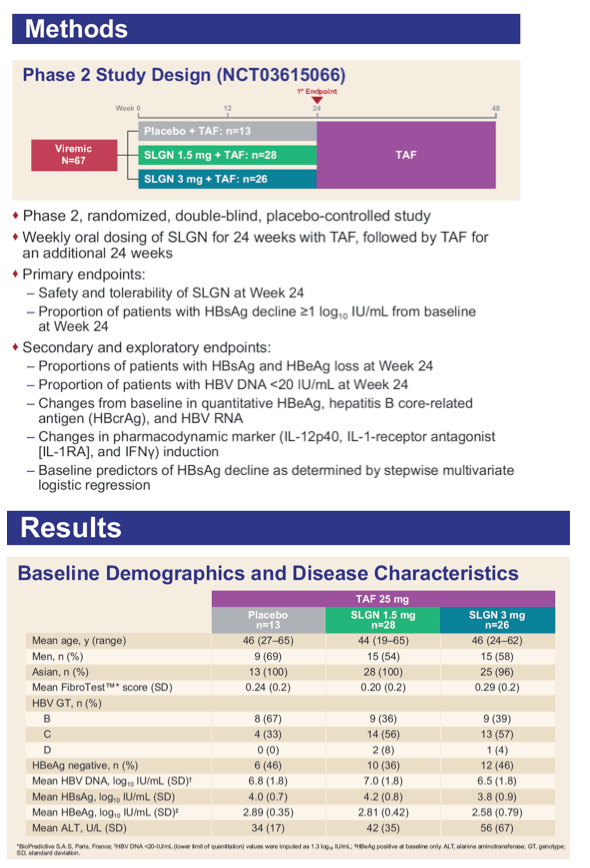



Safety And Efficacy Of 24 Weeks Of Treatment With Oral Tlr8 Agonist Selgantolimod In Viremic Adult Patients With Chronic Hepatitis B A Phase 2 Study
Fections, acute hepatitis B, recipients of immunosuppressive therapy, and transplant recipients The AASLD 18 Hepatitis B Guidance provides a datasupported approach to screening, prevention, diagnosis, and clinical management of patients with hepatitis B It differs from the published 16 AASLD guidelines, which conducted systematic AASLD VBI Vaccines details hep B candidate as it preps filing A twodose vaccine has obvious advantages over a threedose one But in a threedose world where people aren't very good at News > Medscape Medical News > Conference News > AASLD Mixed Outcomes in Tenofovir Trial for Chronic Hepatitis B Emily Willingham About onethird of patients with




sld Cregg




Addressing The Global Burden Of Hepatitis B Virus While Developing Long Acting Injectables For The Prevention And Treatment Of Hiv The Lancet Hiv
Aligos Therapeutics Presents Data on Chronic Hepatitis B, NASH and Hepatocellular Carcinoma Programs at AASLD's The Liver Meeting Digital Experience™ read this article along with other careers information, tips and advice on BioSpaceFeb;71(2) doi /hep Authors Marc G Ghany 1 , Timothy R Morgan 2 , AASLDIDSA Hepatitis C Guidance Panel The AASLD Transplant Hepatology Board Review Course set for August 1516 in Dallas, TX is now an ondemand course This ondemand course will release on Friday, For further questions contact education@aasldorg




sld Vbi Vaccines Details Hep B Candidate As It Preps Filing Fiercebiotech




Easl Assembly Finds A Partner As Hepatitis B Inches Forward Evaluate
Dicerna Announces Positive Updated Data From Phase 1 Trial of RG6346 for Treatment of Chronic Hepatitis B Virus (HBV) Infection at AASLD's The Liver Meeting® Digital Experience™Mission To promote national and international collaboration to promote clinical care and research in the field of the hepatitis B virus (HBV) To improve awareness among health care professionals and the public regarding the prevention of HBV, screening high risk populations for HBV and management of chronic HBV based on AASLD Guidelines Dicerna Announces Updated Phase 1 Data on RG6346 Investigational Treatment for Chronic Hepatitis B Virus to be Presented in LateBreaking Session at AASLD's The Liver Meeting® Digital Experience™ Read full article 1 November , 900 am 5min read Dicerna Pharmaceuticals, Inc (Nasdaq DRNA) (the "Company" or "Dicerna"), a leading developer of




Hbv Drugs In Development Hepatitis Clinical Care Options
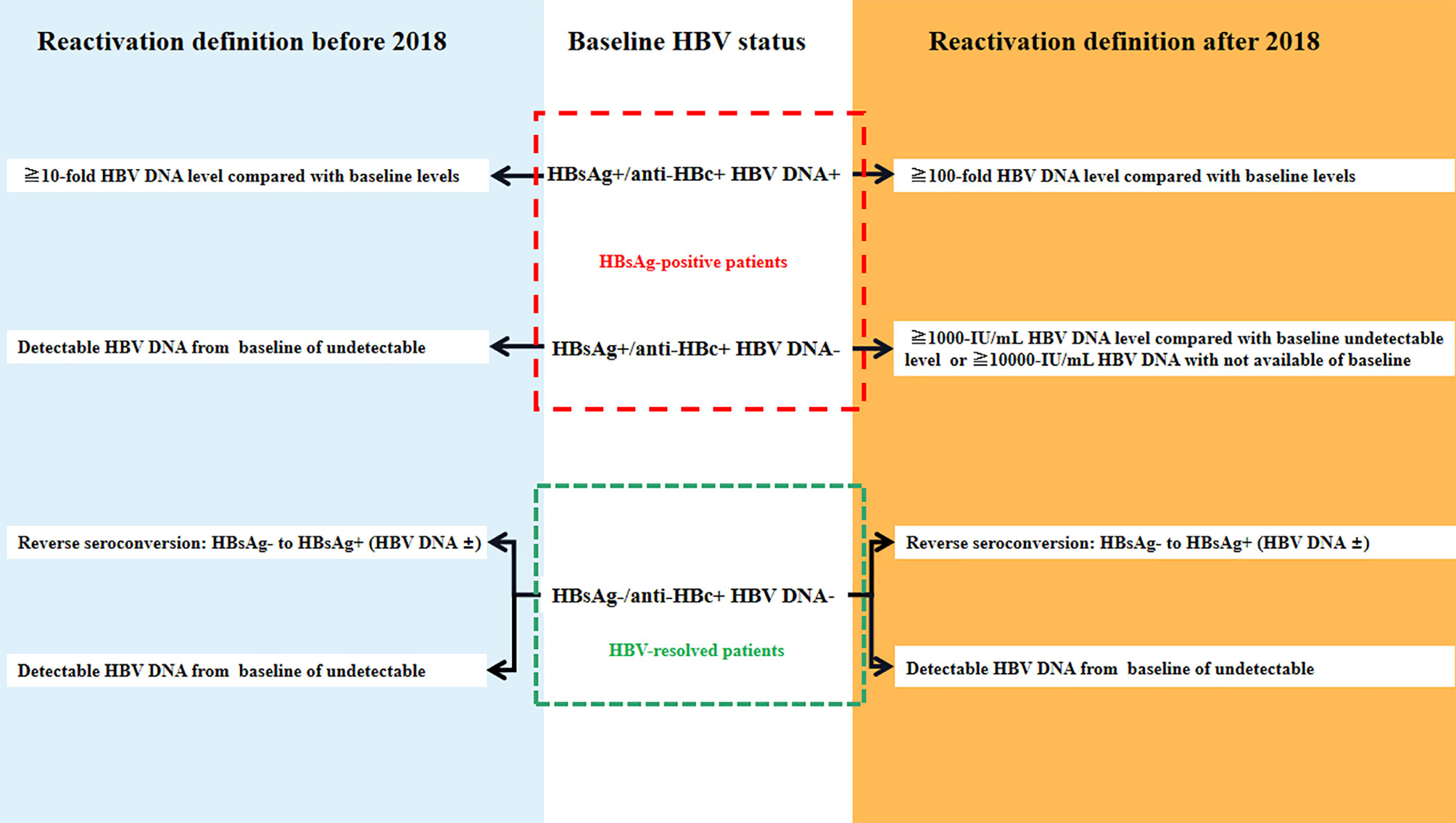



Frontiers Management Of Hepatitis B Virus In Allogeneic Hematopoietic Stem Cell Transplantation Immunology
AASLD Update on Prevention, Diagnosis and Treatment of Chronic Hepatitis B (February 18) ) Hepatitis B Vaccination, Screening, and Linkage to Care Best Practice Advice From the American College of Physicians and the Centers for Disease Control and Prevention (17) Countryspecific Guidelines Some countries with a high prevalence of hepatitis have developed their Worldwide, in hepatocellular carcinoma (HCC) is the seventh most common malignancy and the Diagnosis, and Treatment of Chronic Hepatitis B AASLD 18 Hepatitis B Guidance This document includes recommendations for HCC surveillance in persons with chronic HBV infection Note these recommendations differ from the 18 AASLD Hepatocellular Dicerna Announces Updated Phase 1 Data on RG6346 Investigational Treatment for Chronic Hepatitis B Virus to be Presented in LateBreaking Session at AASLD




Comparison Of The Current International Guidelines On The Management Of Hcc Sciencedirect




Hepatitis B Virus Advances In Prevention Diagnosis And Therapy Clinical Microbiology Reviews
Saturday, Parallel Session Hepatitis B Therapeutics (Established) 900 – 1030 am 900 am Abstract #19 Randomized trial of 192 weeks of tenofovir with or without peginterferon alfa for the first 24 weeks followed by protocolized withdrawal in adults with chronic hepatitis B



2




Clinical Profile And Efficacy Of Antivirals In Hepatitis B Virus Reactivation In Patients With Cancer Receiving Chemotherapy Journal Of Clinical And Experimental Hepatology




Switching From Tenofovir Disoproxil Fumarate To Tenofovir Alafenamide In Virologically Suppressed Patients With Chronic Hepatitis B A Randomised Double Blind Phase 3 Multicentre Non Inferiority Study The Lancet Gastroenterology Hepatology




Hepatitis B Virus Reactivation Potentiated By Biologics Infectious Disease Clinics




American Association For The Study Of Liver Diseases United States Coalition For Global Hepatitis Elimination




Eiger Presents Clinical Trial Results At The Liver Meeting Digital Experience Hepatitis B Foundation




Hepatitis B Virus From Diagnosis To Treatment
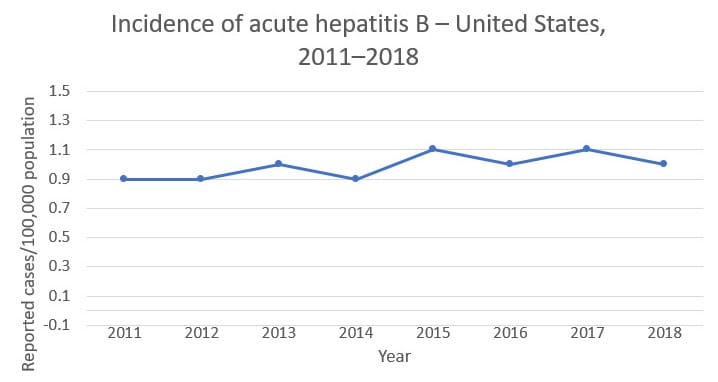



Hepatitis B Questions And Answers For Health Professionals Cdc




Viral Hepatitis Highlights Downloadable Slideset sld Infectious Disease Clinical Care Options




Hepatitis B Virus Persistence And Reactivation The Bmj




Cost Effectiveness Of Antiviral Treatment In Adult Patients With Immune Tolerant Phase Chronic Hepatitis B Gut



sld Guidelines Hepatitis B Reactivation



New Resource Guide To Hepatitis B Management For Primary Care Providers Hepatitis B Foundation



1




Safety And Efficacy Of 24 Weeks Of Treatment With Oral Tlr8 Agonist Selgantolimod In Viremic Adult Patients With Chronic Hepatitis B A Phase 2 Study




Unresolved Issues Of Immune Tolerance In Chronic Hepatitis B Springerlink



Www sld Org Sites Default Files 19 06 Hbvguidance Terrault Et Al 18 Hepatology Pdf



Www sld Org Sites Default Files 19 06 sld 18 Hcc Guidance On Diagnosis 2c Staging And Management Hep 281 29 Pdf




Adding Pegylated Interferon To Tenofovir Improves Odds Of Hbsag Loss In Hepatitis B Patients Aidsmap



Core Concepts Preventing Hbv Perinatal Transmission Prevention Of Hbv Hepatitis B Online



Prevention Of Hbv Recurrence After Liver Transplant A Review



To Hep B Or Not To Hep B Oligonucleotide Therapeutics Society




Xuxwbwrjrjv7m




International Workshop On Hbv Cure Ame




Estimating The Proportion Of People With Chronic Hepatitis B Virus Infection Eligible For Hepatitis B Antiviral Treatment Worldwide A Systematic Review And Meta Analysis The Lancet Gastroenterology Hepatology
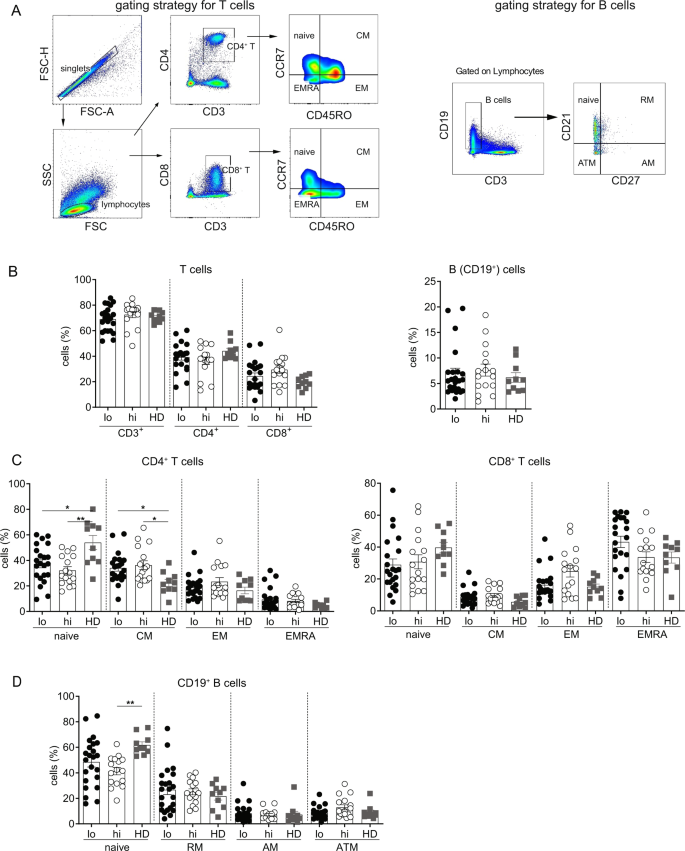



Circulating Serum Hbsag Level Is A Biomarker For Hbv Specific T And B Cell Responses In Chronic Hepatitis B Patients Scientific Reports
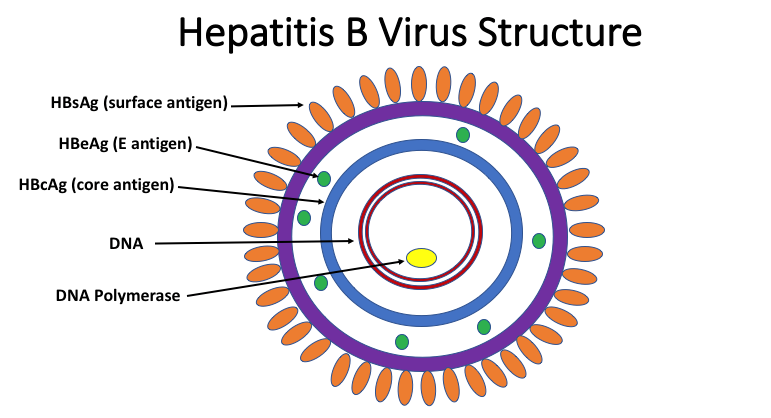



Hepatitis B Serologic Testing Methods




sld Posts Facebook




Core Concepts Hepatitis B Coinfection Co Occurring Conditions National Hiv Curriculum




Hepatitis B Screening Prevention Diagnosis And Treatment American Family Physician




Comparison Of Current Clinical Practice Guidelines For Chronic Download Scientific Diagram




Can More People Stops Nrtis For Hbv Hints From A 48 Week Study




sld Posts Facebook




The Liver Meeting Hepatitis Sessions Coalition For Global Hepatitis Elimination




Hepatitis B And Pregnancy Virologic And Immunologic Characteristics Joshi Hepatology Communications Wiley Online Library
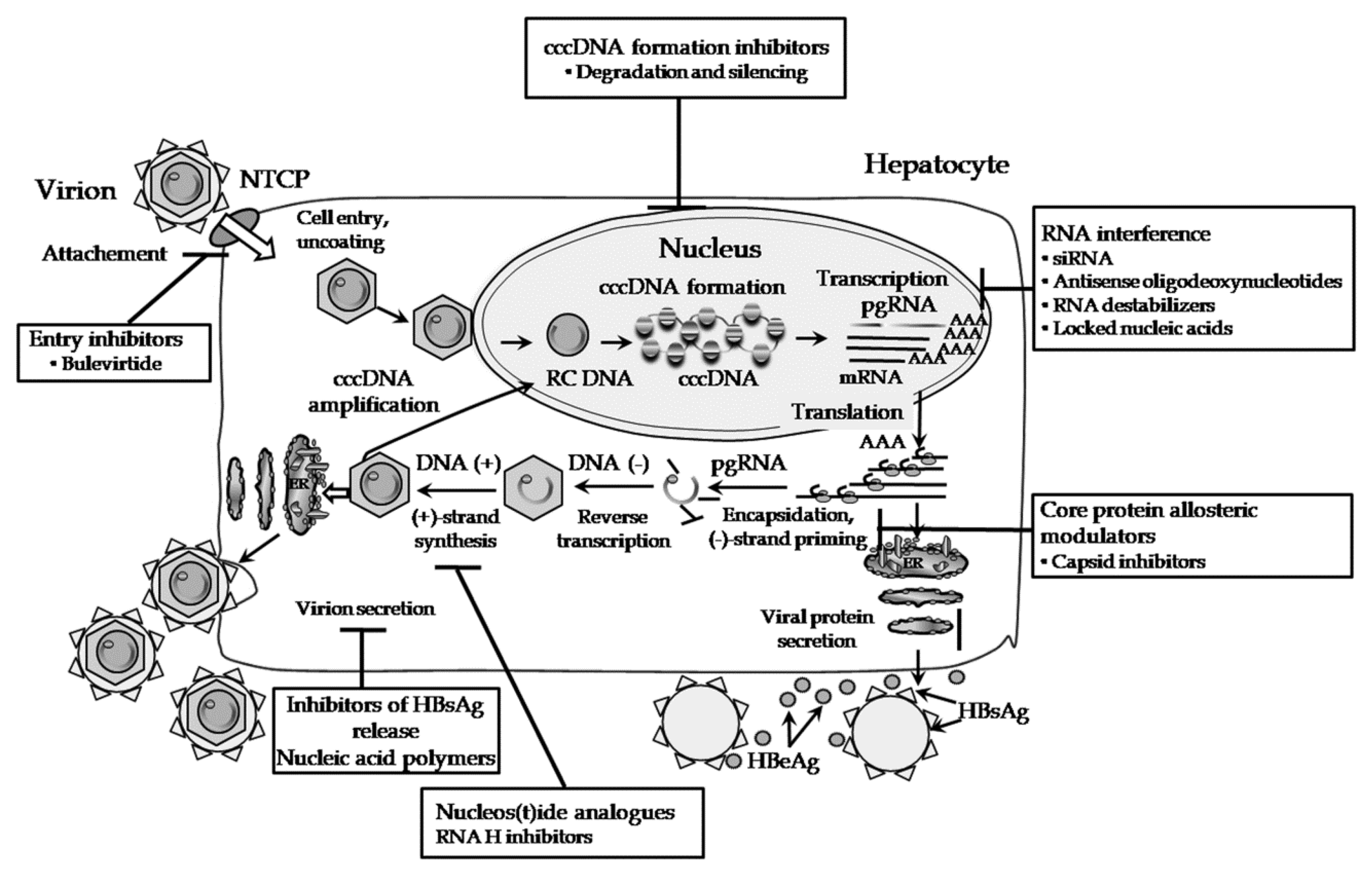



Jcm Free Full Text New Approaches To The Treatment Of Chronic Hepatitis B Html
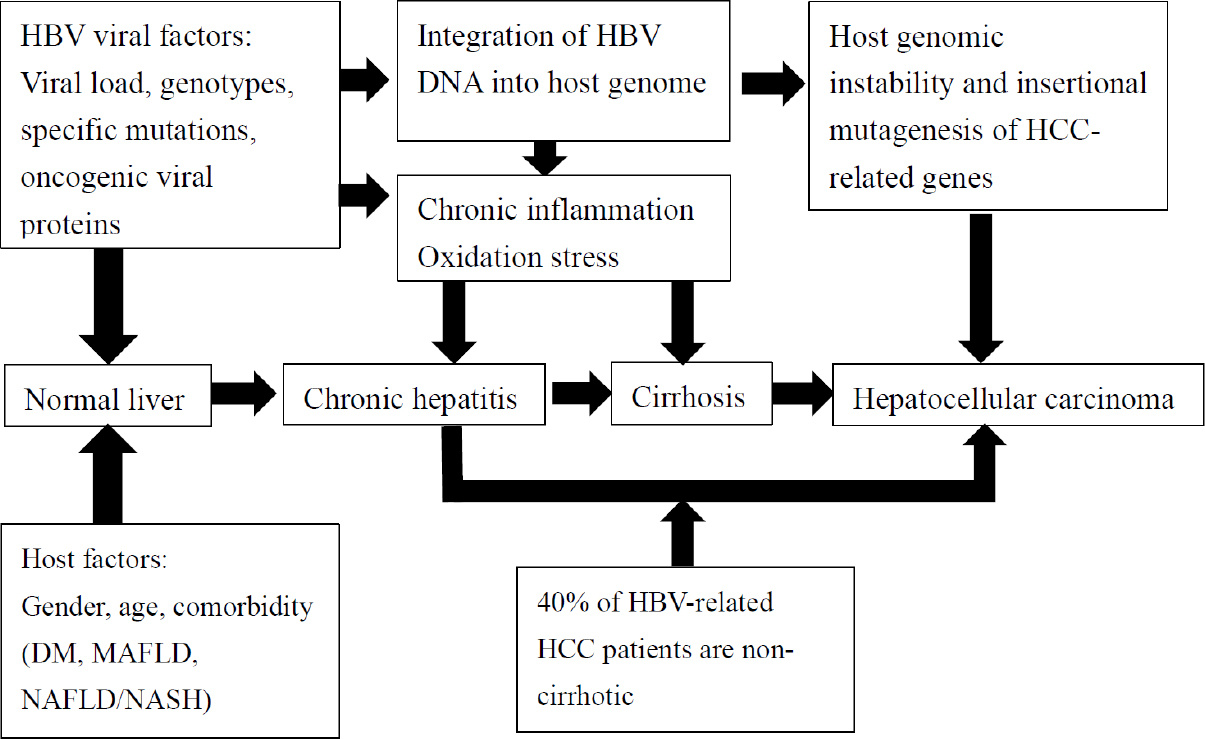



Prevention Of Hepatitis B Virus Related Hepatocellular Carcinoma




Tenofovir During Pregnancy Reduces Risk Of Mother To Child Hepatitis B Virus Transmission Aidsmap



Tlmdx From sld Surfing The Nash Tsunami Podcast Hep Dynamics
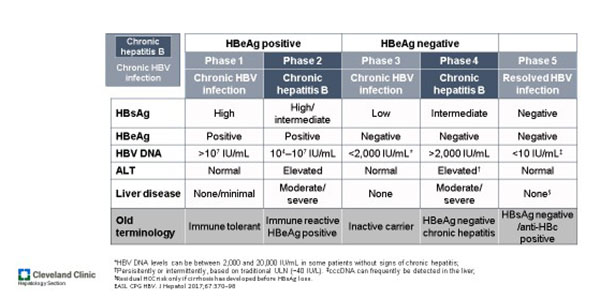



Hepatitis B Cleveland Clinic




Mondaynightibd Hbv Prophylaxis In Ibd Pt1




Pdf Comparison Of Clinical Practice Guidelines For The Management Of Chronic Hepatitis B When To Start When To Change And When To Stop



Having Trouble Reading This Email View It In Your Browser November Contents News From The sld Liver Meeting Which People With Chronic Liver Disease Have Higher Covid 19 Risks Fatty Liver Greatly Increases Vulnerability To Covid 19 In




Hepatitis B Virus And Delta Infection Special Considerations In The Indigenous And Isolated Riverside Populations In The Amazon Region Cabezas Clinical Liver Disease Wiley Online Library
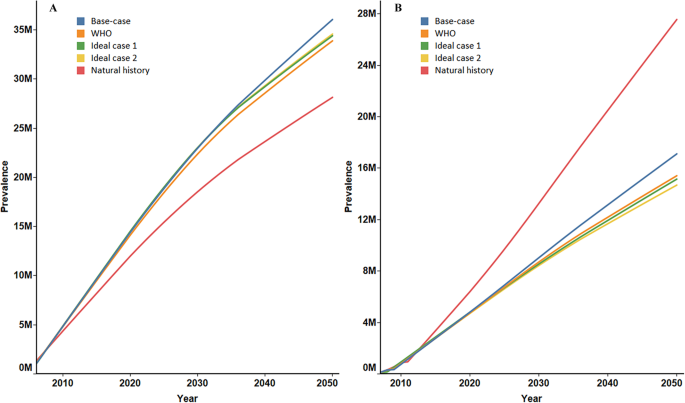



Disease Burden Of Chronic Hepatitis B And Complications In China From 06 To 50 An Individual Based Modeling Study Virology Journal Full Text




Nucleic Acid Polymers Are Effective In Targeting Hepatitis B Surface Antigen But More Trials Are Needed Gastroenterology




Practice Guidelines sld




Guidance For Design And Endpoints Of Clinical Trials In Chronic Hepatitis B Report From The 19 Easl sld Hbv Treatment Endpoints Conference Cornberg Hepatology Wiley Online Library




sld Guidelines Hepatitis B 18 Ppt




Real World Single Center Experience On The Efficacy Of Stopping Long Term Nucleos T Ide Analog Therapy In Patients With Chronic Hepatitis B
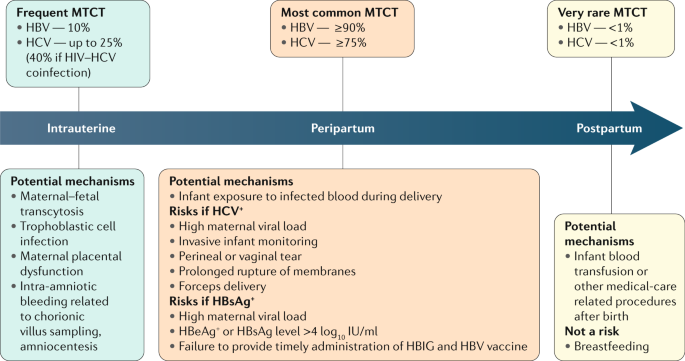



Viral Hepatitis And Pregnancy Nature Reviews Gastroenterology Hepatology




Practice Guidelines sld




Kmt Hepatech Check Out Hbv Related Papers Published In Where The Pxb Mouse And Pxb Cells Were Used In Studies Tlmdx Hepatitis Liver Drugdevelopment Drugdiscovery T Co Hxigqgycuw T Co Edvvfhaark



Infohep Few Countries Will Meet Target For Reducing Viral Hepatitis Deaths




Hepatitis B Secondary Prevention Wikidoc



Www Mdpi Com 1873 149x 27 1 2 Pdf



Biomarkers Associated With Alt Flares In Hbsag Decline In Patients With Chronic Hepatitis B Cellcarta



Q Tbn And9gctmrmzsbh1zirvbxaxhr5ctfegn7 Ai Lnode9rvfxy4qoj Rhh Usqp Cau
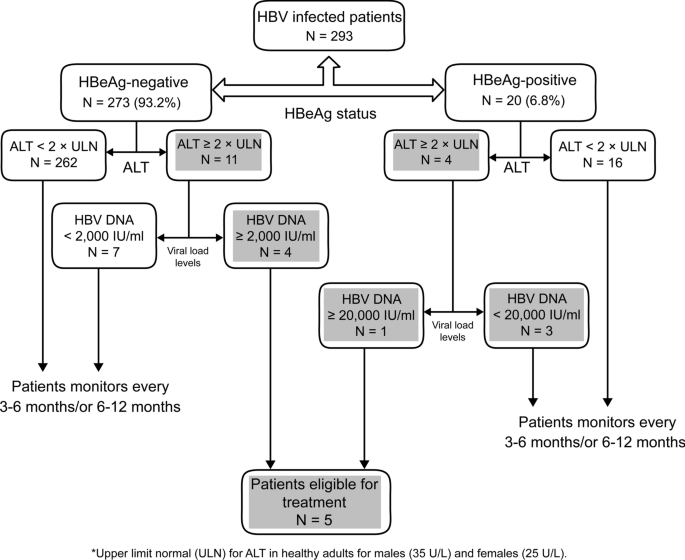



Prevalence Of Chronic Hepatitis B Phases In Eritrean Patients A Laboratory Based Cross Sectional Study Bmc Gastroenterology Full Text



Q Tbn And9gcrkzonwgtzcedgmj9ogppxv0l Dmihh8fgwf3fsd1z79lrxfvfo Usqp Cau




Hepatitis B Amboss




Hepatitis B Related Outcomes Following Direct Acting Antiviral Therapy In Taiwanese Patients With Chronic Hbv Hcv Co Infection Sciencedirect




Hepatitis B Screening Prevention Diagnosis And Treatment American Family Physician




Easl 17 Clinical Practice Guidelines On The Management Of Hepatitis B Virus Infection Journal Of Hepatology




Jhoxplobwhaqum




Abi H0731 A New Hepatitis B Drug Under Development In The First Half Of 21 Registered For Trial In China Inews




Hepatitis B Virus Whats New Easl Treatment Guidelines




Hepatitis B Infection Gutsandgrowth




Practice Guidelines sld



Www sld Org Sites Default Files 04 Covid19 Clinicalinsightsupdates 416 Pdf
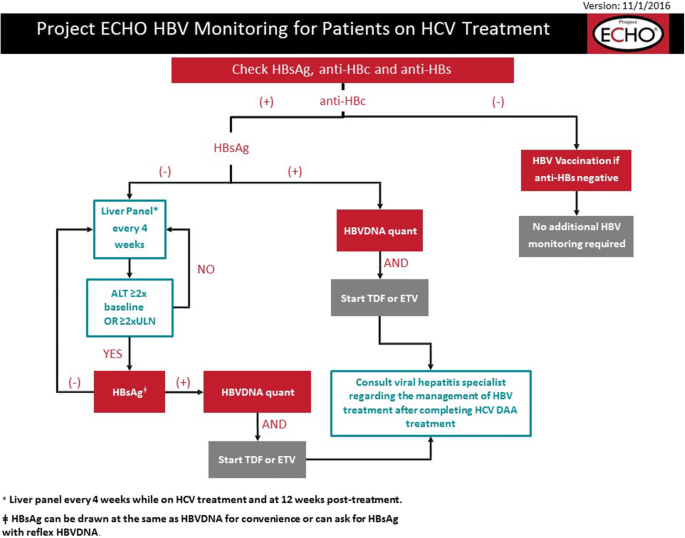



Hepatitis B Core Antibody Role In Clinical Practice In Springerlink



2




One World One Pandemic Many Guidelines Management Of Liver Diseases During Covid 19 Gut




Easl 17 Clinical Practice Guidelines On The Management Of Hepatitis B Virus Infection Journal Of Hepatology




sld Updates Chronic Hepatitis B Recommendations Practice Guidelines American Family Physician




Easl 17 Clinical Practice Guidelines On The Management Of Hepatitis B Virus Infection Journal Of Hepatology




sld Recommendations For Treatment Of Chronic Hepatitis B Download Table




Practice Guidelines sld



0 件のコメント:
コメントを投稿